|
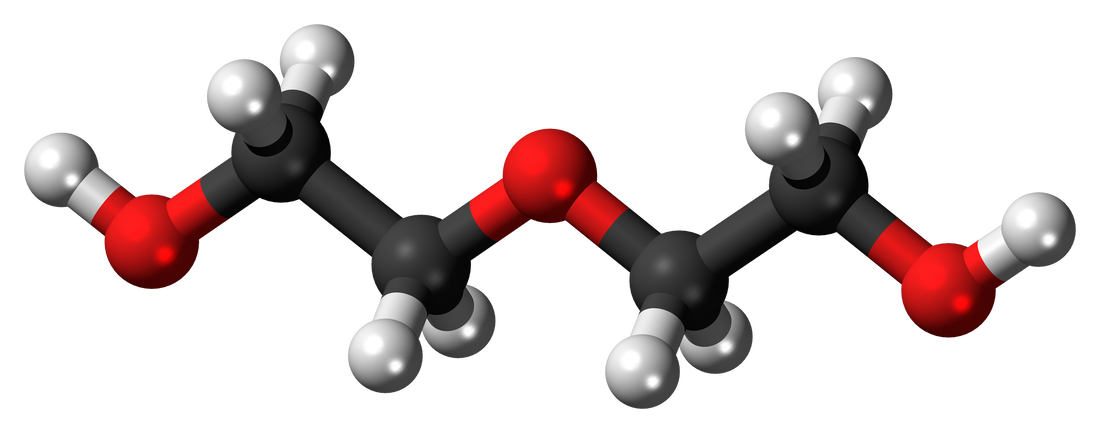
Diethylene Glycol is a colorless, odorless, practically odorless organic compound with the chemical formula HOCH2CH2O. It is hygroscopic, has a slightly sweet taste, and is a four-carbon dimer of ethylene glycol. Despite its relatively low toxicity, it is still widely used as a solvent in the chemical industry.
It has been linked to numerous cases of mass poisoning. While it is widely available in household products, there is still no comprehensive literature review of its toxicology. DIEG is a toxic compound with a high mortality rate. So, what are the application of Diethylene Glycol? It is a common ingredient in many household products, including cleaning supplies, textiles, and adhesives. Its properties make it a popular choice for cosmetics and other industrial uses. Its properties make it a useful chemical. It can be extremely toxic, and contact with it can lead to skin irritation, severe inflammation, and liver poisoning. It is used to make cigarettes. It is metabolized in the liver in the same NAD-dependent pathway as ethanol. It is then converted into 2-hydroxyethoxyacetic acid by aldehyde dehydrogenase. As a by-product of plastic production, it is highly useful and can be stored in many containers. Its flammability rating is one and its flashpoint is 143 degrees Celsius. It is not regulated for transportation, but it is harmful if ingested or inhaled. The chemical vapors of this chemical can also cause severe burns. In addition to its wide range of applications, it can be found in petroleum solvent extraction, printing ink, drywall joint compounds, and dental floss. Its lowest toxicity is found in triethylene glycol. The toxicity of Diethylene Glycol is not yet fully understood. The chemical is a product of ethylene glycol metabolism. There are other metabolites that are involved in its metabolism. Some studies suggest that DEG toxicity is different from ethylene glycol toxicity. Its metabolism is the underlying factor in toxic effects. It is not fully understood how toxic DEG is, but it is worth mentioning that the toxicity is distinct from ethylene glycol. The chemical is commonly found in foods and beverages. However, it is a lesser-known poison than ethylene glycol. Despite this, DEG has been linked to several poison outbreaks in history. The 1937 Massengill antibiotic poisoning was the catalyst for the establishment of the U.S. Food and Drug Administration. It has also been linked to mass outbreaks in Haiti (1996), Bangladesh (1990), and Panama (2006). Despite this high level of toxicity, this chemical is widely used in many industries. In a recent study, rats were administered 2 g/kg DEG, at a low dose, and 10 g/kg at a high dose. Blood was collected over 48 hours. At high doses, the rats suffered metabolic acidosis, increased BUN and creatinine, and severe renal damage. Although the high dose did not result in any significant kidney damage, the low dose did result in marked kidney necrosis. In addition to these effects, DEG was also linked to respiratory depression and impaired motor function.
0 Comments
Leave a Reply. |
|